一项新的研究可能有助于解释自闭症患者的嗅觉障碍
纽约理工学院骨科医学院(NYITCOM)的一项新研究可以帮助解释自闭症患者的嗅觉是如何受到影响的。
自闭症患者有一种“坚持千篇一律”的倾向,他们经常避免接触不熟悉的元素,包括可能影响他们生活质量的新气味和新食物。虽然许多研究都集中在自闭症的行为特征上,但还需要更多的研究来帮助解释它的感官方面。
现在,NYITCOM生物医学科学助理教授冈萨洛·奥塔祖博士领导的一项研究发表在杂志上自然通讯他分析了自闭症小鼠模型,并报告了负责嗅觉的神经过程的差异。
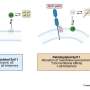
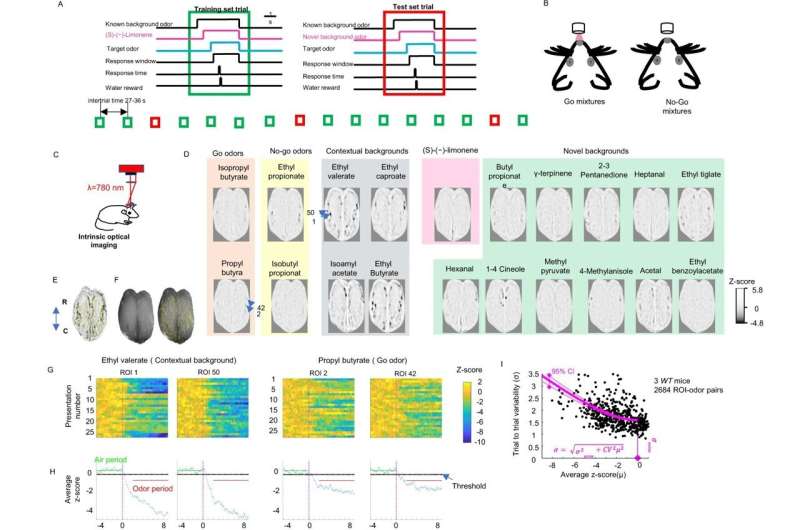
研究小组训练了两组小鼠——一组与自闭症相关的基因发生突变(CNTNAP2敲除小鼠),另一组神经正常的小鼠——来识别熟悉的气味。当他们成功地识别出目标气味时,奖励给老鼠一口水。两组人都成功地确定了目标。
然后,老鼠被赋予了一项更具挑战性的任务:在背景中引入不熟悉的气味时,识别目标气味。作为一名电气工程师,Otazu将这项任务比作互联网验证码,它要求人们在繁忙或模糊的背景中直观地识别字母和数字。虽然神经正常的小鼠能够“过滤”出新的背景气味并识别出目标气味,但CNTNAP2敲除小鼠却很难做到这一点。
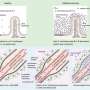
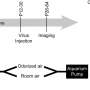
为了更好地理解处理错误发生在CNTNAP2敲除小鼠的大脑中,研究人员将其可视化神经活动在每个动物的输入嗅球前额叶是大脑最初处理气味的部分。一种称为本征光学成像的成像技术被用于可视化嗅球表面附近的神经活动。在这里,“气味信号”被传输到大脑的其他部位进行进一步处理,在大脑计算气味的过程中发挥着关键作用。
然而,CNTNAP2敲除小鼠和神经正常小鼠之间的输入信号非常相似。这表明,自闭症模型中的气味处理在稍后的步骤中受损——在嗅球输入的信号处理之后。当研究人员对大脑识别陌生背景下目标气味的过程进行“逆向工程”时,这一发现也得到了印证。
利用机器学习这是一种使用算法来复制大脑过程的人工智能形式,该团队将嗅球输入信号应用于一种复杂的算法,该算法与神经典型小鼠的高性能相匹配。正常的老鼠过滤掉新的背景气味并识别目标,但CNTNAP2的这种复杂处理受到了损害基因敲除小鼠.
“我们推测,自闭症小鼠模型中的嗅球可能更容易被处理新的背景气味所淹没,”Otazu说。“这些发现说明了为什么更多有关自闭症感官方面的研究如此重要。通过记录自闭症小鼠模型中的神经过程,我们的发现可能有助于解释患有自闭症的人类大脑回路自闭症总有一天会改善这些人的生活质量。”
更多信息:Yan Li等,小鼠在新嗅觉环境中的强大气味识别,自然通讯(2023)。DOI: 10.1038 / s41467 - 023 - 36346 - x