针对端粒对肺癌可能是一种有效的治疗策略,根据研究
健康细胞只能把有限的次数在有机体的一生。相反,肿瘤细胞是不朽的:他们无限增殖和失控,这是癌症的定义特征。
端粒和端粒酶的研究人员小组CNIO(西班牙国家癌症研究中心),由Maria Blasco首次研究了针对治疗肺癌的可能性端粒结构,保护染色体的末端和无限期的条件决定了细胞的分裂能力。
解释的结果,研究人员在《华尔街日报》细胞死亡与分化,表明,事实上,针对端粒”可能是一种有效的治疗策略”对非小细胞肺癌,负责大部分肺癌患者的死亡率。Sergio Pineiro工作作为第一作者,接受者的博士后合同西班牙与癌症协会(AECC)。
“把永生从癌症细胞是一种治疗策略,尚未利用对抗癌症,”Maria Blasco说。
专注于肿瘤微环境
肺癌是癌症死亡的主要原因之一。当前的治疗和诊断晚期的长期无效意味着只有五分之一的病人存活超过5年。具体地说,非小细胞肺癌的85%负责肺癌相关的死亡。
现在的工作发表关注所谓的肿瘤微环境,这是一组细胞和肿瘤周围的因素在癌症的发展起着至关重要的作用和对治疗的反应。
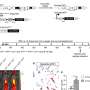
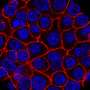
研究人员分析了端粒功能失调的影响。同时,他们研究了端粒酶的作用缺乏在非小细胞肺癌的细胞微环境,端粒酶被修复端粒的酶。
端粒酶缺乏症
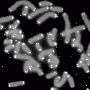
端粒是染色体末端的一种蛋白质结构位于。在每一个细胞分裂,端粒缩短,直到一定数量的分歧后,缩短变得过度,细胞停止分裂。这发生在健康的细胞,但不是大多数肿瘤细胞。
端粒酶表达在人类肿瘤的90%被重新激活。由于端粒酶的作用,肿瘤细胞的端粒维持一个最低功能长度,这允许他们将下去。
CNIO研究人员研究了发生了什么当他们引起了端粒酶赤字肺肿瘤微环境。他们还故意损害了他们的端粒,使用复合6-thio-dG。
“这是第一次的参与端粒酶和功能失调的端粒在肺肿瘤微环境调查,”塞吉奥Pineiro解释说,目前西班牙国家研究委员会(CSIC)在洛杉矶的里奥哈。
损坏的端粒抑制肿瘤
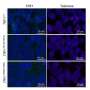
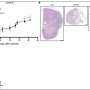
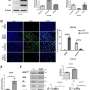
端粒酶缺乏症和功能失调的端粒延缓肿瘤进展。研究人员观察到减少肿瘤植入和肺血管化,同时增加的脆弱性肿瘤DNA损伤和细胞死亡。肿瘤细胞增殖和炎症也减少,免疫系统的抗肿瘤反应增强。
正如作者在写细胞死亡与分化,“我们地址第一次叔的含义(端粒酶)和功能失调的端粒在肺肿瘤微环境。我们的研究结果表明,针对端粒可能是一种有效的治疗策略非小细胞肺癌。"
更多信息:塞吉奥Pineiro-Hermida et al,端粒酶缺陷和功能失调的端粒在非小细胞肺癌肺肿瘤微环境影响肿瘤进展小鼠模型和patient-derived异种移植,细胞死亡与分化(2023)。DOI: 10.1038 / s41418 - 023 - 01149 - 6